Podcast 124 - Arterial Line Placement in Critical Care Transport w/ Michael Lauria
- Colin Standifird, BA, FP-C; Michael Wasserman, MD; and Michael Lauria, MD, NRP, FP-C”
- Jun 25, 2021
- 8 min read

Written by: Colin Standifird, BA, FP-C; Michael Wasserman, MD; and Michael Lauria, MD, NRP, FP-C”
Invasive arterial blood pressure (IABP) monitoring techniques have enjoyed a rich history of use throughout the mid-to-late 20th century in the peri-operative setting and are now a standard of care intensive care units. While there are a variety of IABP monitoring options, one of the most common techniques is percutaneous radial arterial catheterization. Although monitoring of radial arterial lines is a widely accepted skill in the critical care transport (CCT) world, placement by CCT providers is less common. Concerns over safety and logistical management have contributed to the perceived difficulty in arterial lines; however, this article aims to demonstrate that arterial lines can be placed safely and effectively in the pre-hospital setting.
CCT personnel regularly transport critically ill patients and perform interventions such as initiation of vasopressors and antihypertensives. They also manage patients that might benefit from tight blood pressure control, such as strokes, spinal cord injury patients, traumatic head injuries, shock, hypertensive emergencies, or patients who are peri-arrest.2 These patients are transported from the field as well as from rural, resource-limited hospitals where initial stabilization of the patient has occurred and long distances need to be covered before arrival at a tertiary or quaternary care facility.
The mainstay of blood pressure monitoring in CCT (and largely in the intra-hospital setting as well) is through the use of disposable, oscillometric, non-invasive blood pressure (NIBP) cuffs. Oscillometric BP cuffs function by inflating the cuff bladder to occlude arterial flow and then slowly deflate while an internal pressure sensor detects oscillations in blood flow as the cuff is deflated; the maximum oscillation detected corresponds to the patient’s MAP, and systolic and diastolic pressures are then estimated through the use of the BP cuff manufacturer's proprietary algorithm.3
Despite their ease of use, the inaccuracy of NIBP monitoring techniques is well documented in the scientific and medical literature. Moreover, in terms of management during CCT transport, the error in NIBP readings can be clinically unacceptable. In a meta-analysis of 28 studies including 919 adult ICU and OR patients, Kim et al found a standard deviation of the bias between invasive arterial (IABP) and NIBP of 8.4 with a significant number of measurements with more than 10 mmHg difference. This did not meet the Association for the Advancement of Medical Instrumentation acceptable standard for clinical validation of the NIBP devices. Ribezzo et al used a randomized crossover study design to evaluate the agreement between IABP and NIBP measurements in critically ill ICU patients (septic shock, hemorrhagic shock, major trauma, and post-cardiac arrest patients) and found that, on average, the NIBP measurements were off by 10.8 mmHg.5 Furthermore, nearly a quarter of the patients in this study demonstrated NIBP readings that were recorded being more than 20 mmHg difference from the IABP. In a direct comparison of IABP to NIBP measurement, McMahon et al found that 45.8% of systolic blood pressure readings were outside of their institution’s acceptable limit of 10 mmHg difference with NIBP readings documented as up to 34.4 mmHg off the IABP reading.6 Perhaps most concerning regarding the management of critical patients, authors have noted the largest differences at extremes of blood pressure when differences are perhaps most important. Specifically, NIBP will overestimate low blood pressure and underestimate very high blood pressure.
There are a variety of factors that influence the oscillations which can provide falsely elevated/decreased readings, including movement during transport while the blood pressure cuff is being inflated/deflated, arrhythmia, arteriosclerosis, low or fluctuating cardiac output, hypotension (<100 mm Hg), hypertension (>180 mm Hg), cuff size, upper arm size, diabetes mellitus, critical illness, and increased pulse pressure.
The lack of reliability of non-invasive blood pressure measurements suggests that, in certain critical patients being transported, IABP would be prudent. Therefore, it may be reasonable for CCT personnel to place radial arterial lines for more accurate blood pressure monitoring and titration of therapies prior to or during transport.
Brief Review of Concept and Equipment
Percutaneous placement of a radial arterial catheter is accomplished through the use of the Seldinger Technique (catheter inserted over a guidewire) with or without ultrasound guidance. Most commercially available radial arterial line kits utilize a 20 gauge needle for percutaneous access, a flexible spiral guidewire, and a short (35mm) arterial catheter.
The basic equipment necessary to perform this procedure from a commercially available kit are detailed below and visually depicted in Figure 1. A more compact kit carried by a transport team is depicted in Figure 2.
1.) Short (35mm) 20 gauge arterial catheter with a guidewire
2.) Sterile gloves, hair cap, mask
3.) Chloraprep Scrub
4.) Sterile connector tubing to attach to the monitoring system
5.) 3.0 silk straight suture or tape
6.) Clear bio-occlusive dressing
7.) Arterial monitoring system with pressure transduction tubing, pressure bag, and 1 liter lactated ringer’s or normal saline bolus
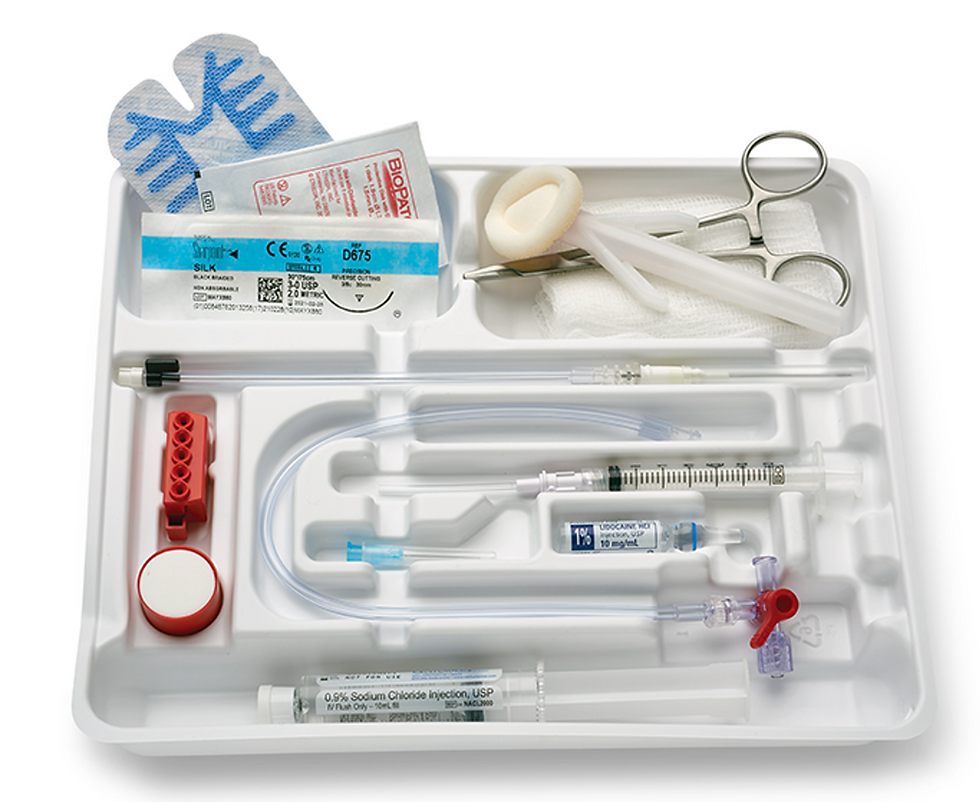
Figure 1. Detailed picture of necessary equipment in pre-made commercially available kit
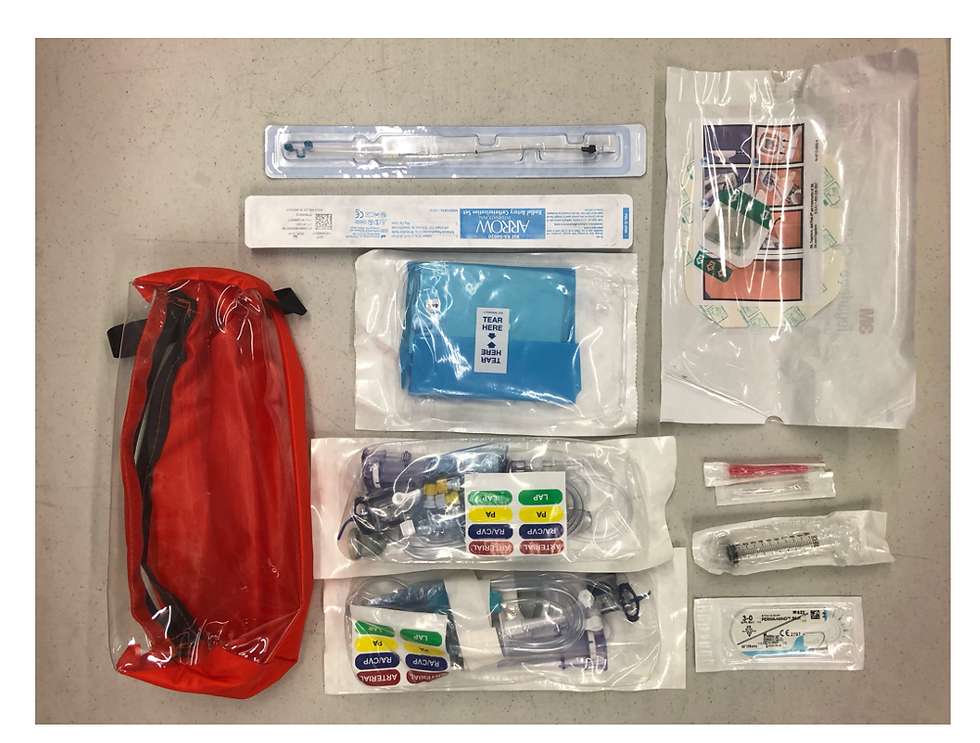
Figure 2. Smaller, compact transport arterial line kit with the addition of arterial line tubing.
The overall safety profile of radial arterial line monitoring is well documented. Absolute contraindications to the procedure are primarily related to conditions that predispose to poor circulation in the right or left distal upper extremity, including full-thickness burns over the forearm, history of vasculitis (thromboangiitis obliterans), and Raynaud Syndrome; while relative contraindications include systemic anticoagulation and/or uncontrolled coagulopathy. Complications of the procedure are low and rarely result in significant morbidity. Scheer et al. conducted a recent review of the most common complications and found that the most frequent adverse events were, in order of decreasing frequency, temporary radial artery occlusion (19.7%), hematoma (14.4%), local infection (0.72%), hemorrhage (0.53%), systemic bacteremia (0.13%), and permanent ischemic damage or pseudoaneurysm (0.09%).
Arterial line placement is most widely used in the perioperative setting, intensive care units, and emergency departments. Despite the many indications, arterial line catheterization is not commonly performed by CCT or prehospital services. However, there are some services around the world the anecdotally suggest the utility of the procedure. One physician-staffed EMS agency published data and noted that 115 arterial lines were placed with an 83.9% success rate. From the decision to monitoring invasive blood pressures took 5 minutes on average with cannulation times taking only 2 minutes of that time. This study concluded that the placement of arterial lines is feasible in the CCT or prehospital setting and does not delay or complicate patient care.
Research shows that with proper training non-physician insertions of invasive lines can be accomplished safely. In a study done at Melbourne metropolitan ICU, 21 nurses were trained in radial artery cannulation. 107 patients underwent cannulation attempts and there were no reports of negative events. In another study, a group of respiratory therapists inserted more than 500 catheters. These arterial lines were left in the patients between 1 and 20 days. Of these insertions, there was only one patient (0.2%) with a serious complication and a less than 5% superficial infection rate.12 These rates of complication are well within the documented complication rates of invasive line placements by other providers.
Since arterial cannulation by non-physicians with special training is safe and there is a reasonable utility of the intervention in transport, it may be feasible to implement this intervention in non-physician transport programs. Furthermore, recent developments in CCT education and training may make arterial line placement and IABP monitoring more common. Ultrasound-guided radial arterial line placement has been shown to decrease time to cannulation, decrease attempts, and limit the number of sites attempted for cannulation.13 As it turns out, many transport services have completed extensive point of care ultrasound training that allows non-physicians to guide treatment and assist in procedures.14 Ultrasound is both feasible and clinically helpful in CCT and prehospital emergency care with comprehensive protocol development and effective training. Therefore, training CCT personnel to use ultrasound for arterial line placement may further increase safety and enhance feasibility.
While NIBP monitoring is convenient in the pre-hospital setting, its use in critically ill patients should be carefully considered given documented inaccuracies. Countless therapeutic patient interventions are based on blood pressure parameters, and the pre-hospital paramedic, nurse, and physician should strongly consider the use of IABP monitoring, especially during critical care transport. Radial artery cannulation for arterial blood pressure monitoring is a safe and effective procedure that can reasonably be performed in the pre-hospital setting by both physicians and non-physicians. CCT teams should consider clinical guidelines that outline indications and training to safely implement this as a clinical skill.
Competency Checklist and CPG with permission from Lifeguard.
References
Wildner G, Pauker N, Archan S, Gemes G, Rigaud M, Pocivalnik M, Prause G. Arterial line in prehospital emergency settings – a feasibility study in four physician-staffed emergency medical systems. Resuscitation. 2011; 82(9): 1198-1201.
2. Cecconi M, De Backer D, Antonelli M, Beale R, Bakker J, Hofer C, et al. Consensus on circulatory shock and hemodynamic monitoring. Taskforce of the European Society of Intensive Care Medicine. Intens Care Med. 2014; 40(12): 1795-1815
Kirkland L. (2009, October). How trustworthy is automated noninvasive blood pressure monitoring? ACP Hospitalist. Published October 2009. Retrieved May 5, 2021. Available at: https://acphospitalist.org/archives/2009/10/traps.htm
Kim S, Lilot M, Sidhu KS, Rinehart J, Yu Z, Canales C, Cannesson M. Accuracy and precision of continuous noninvasive arterial pressure monitoring compared with invasive arterial pressure. Anesthesiology. 2014; 120(5): 1080-1097.
Ribezzo S, Spina E, Di Bartolomeo S, Sanson G. Noninvasive techniques for blood pressure measurement are not a reliable alternative to direct measurement: A randomized crossover trial in the ICU. Sci World J. 2014; 2014; 1-8.
McMahon N, Hogg LA, Corfield AR, Exton AD. Comparison of non-invasive and invasive blood pressure in aeromedical care. Anaesthesia. 2012; 67(12): 1343-1347.
Gourdeau M, Martin R, Lamarche Y, Tetreault L. Oscillometry and direct blood Pressure: A Comparative clinical study during deliberate hypotension. Can Anaes Soc J. 1986; 33(3): 300-307.
Bur A, Herkner H, Vlcek M, Woisetschläger C, Derhaschnig U, Delle Karth G, Hirschl M. M. Factors influencing the accuracy of oscillometric blood pressure measurement in critically ill patients. Crit Care Med. 2003; 31(3): 793-799.
Tiru B, Bloomstone JA. Radial artery cannulation: A review article. J Anes Clin Res. 2012; 03(05): 1-6.
Scheer BV, Perel A, Pfeiffer UJ. Clinical review: Complications and risk factors of peripheral arterial catheters used for haemodynamic monitoring in anaesthesia and intensive care medicine. Crit Care. 2002; 6:199–204.
Chee BC, Baldwin IC, Shahwan-Akl L, Fealy NG, Heland MJ, Rogan JJ. (2011). Evaluation of a radial artery cannulation training program for intensive care nurses: a descriptive, explorative study. Aus Crit Care. 2011; 24(2): 117-125.
Gronheck C, Miller EL. Nonphysician placement of arterial catheters: experience with 500 insertions. Chest. 1993; 104(6): 1716-1717.
Shiver S, Blaivas M, Lyon M. A prospective comparison of ultrasound-guided and blindly placed radial arterial catheters. Acad Emerg Med. 2006; 13(12): 1275-1279.
14. Taylor J, McLaughlin K, McRae A, Lang E, Anton A. Use of prehospital ultrasound in North America: a survey of emergency medical services medical directors. BMC Emerg Med. 2014; 14: 6.
15. Bush M. Portable ultrasound in pre-hospital emergencies: a feasibility study. Acta Anaes Scand. 2006; 50(6): 754-758.
Rudolph SS, Sørensen MK, Svane C, Hesselfeldt R, Steinmetz J. Effect of prehospital ultrasound on clinical outcomes of a non-trauma patients−a systematic review. Resuscitation. 2014; 85: 21–30.
17. O'Dochartaigh D, Douma M, MacKenzie M. Five-year retrospective review of physician and non-physician performed ultrasound in a Canadian critical care helicopter emergency medical service. Prehosp Emerg Care. 2017; 21: 24–31.
18. Cover M, Tafoya C, Long B, Cranford J, Burkhardt J, Huang R, Theyyunni N, Bassin B, Lowell M, Kessler R. Creation of a flight nurse critical care ultrasound program. Air Med J. 2019; 38: 266-272.
19. Meadley B, Olaussen A, Delorenzo A, Roder N, Martin C, St. Clair T, et al. Educational standards for training paramedics in ultrasound: a scoping review. BMC Emerg Med. 2017; 17: 18.
20. Yates JG, Baylous D. Aeromedical ultrasound: the evaluation of point-of-care ultrasound during helicopter transport. Air Med J. 2017; 36: 110–115.
Bøtker MT, Jacobsen L, Rudolph SS, Knudsen L. The role of point of care ultrasound in prehospital critical care: a systematic review. Scand J Trauma Resus Emerg Med. 2018; 26(1): 51.





